 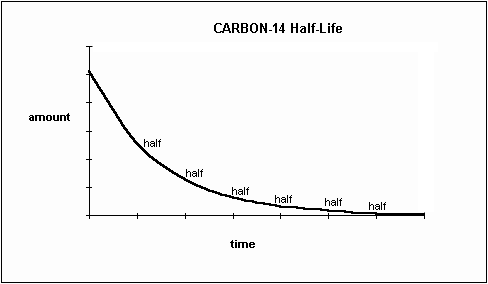
When an organism dies, it stops taking in carbon-14, so the ratio of carbon-14 to carbon-12 in its remains, such as fossilized bones, will decline as carbon-14 decays gradually to nitrogen-14 2 ^2 2 squared. carbon content of a product using radiocarbon (carbon-14). They have the same number of protons and electrons and they are chemically similar. Identify two ways in which carbon-12, carbon-13, and carbon-14 are alike. Carbon-14 has two more neutrons than carbon-12. Carbon-14 is radioactive, carbon-12 is not. As animals eat the plants, or eat other animals that ate plants, the concentrations of carbon-14 in their bodies will also match the atmospheric concentration. differences between the two carbon content types and why each needs to be treated differently. PLAY Identify two differences between carbon-12 and carbon-14. As plants pull carbon dioxide from the air to make sugars, the relative amount of carbon-14 in their tissues will be equal to the concentration of carbon-14 in the atmosphere. 
These forms of carbon are found in the atmosphere in relatively constant proportions, with carbon-12 as the major form at about 99%, carbon-13 as a minor form at about 1%, and carbon-14 present only in tiny amounts 1 ^1 1 start superscript, 1, end superscript. For example, carbon is normally present in the atmosphere in the form of gases like carbon dioxide, and it exists in three isotopic forms: carbon-12 and carbon-13, which are stable, and carbon-14, which is radioactive. the isotopic carbon atom that comprises 99 percent of naturally occurring carbon, and that since 1961 has been used as the standard for atomic weight by representing a unit of 12.00000. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
